Nội dung toàn văn Circular No. 28/2012/TT-BKHCN providing for announcement of standard conformity and announcement
|
THE
MINISTRY OF SCIENCE AND TECHNOLOGY |
SOCIALIST REPUBLIC OF
VIET NAM |
|
No: 28/2012/TT-BKHCN |
Hanoi, December 12, 2012 |
CIRCULAR
PROVIDING FOR ANNOUNCEMENT OF STANDARD CONFORMITY AND ANNOUNCEMENT OF TECHNICAL-REGULATION CONFORMITY AND METHOD TO ASSESS CONFORMITY WITH STANDARDS AND TECHNICAL REGULATIONS
Pursuant to the Law on Standards and Technical Regulations dated June 29, 2006;
Pursuant to the Law on product and goods quality dated November 21, 2007;
Pursuant to the Government’s Decree No.127/2007/ND-CP dated August 01, 2007, detailing implementation of a number of Articles of Law on Standards and Technical Regulations;
Pursuant to the Government’s Decree No.132/2008/ND-CP dated December 31, 2008, detailing implementation of a number of Articles of Law on product and goods quality;
Pursuant to the Government’s Decree No.28/2008/ND-CP dated March 14, 2008, defining the functions, tasks, powers and organizational structure of the Ministry of science and technology;
At the proposal of General Director of Directorate for Standards, Metrology and Quality;
The Minister of science and technology provides for announcement of standard conformity and announcement of technical-regulation conformity and method to assess the conformity with standards and technical regulations.
Chapter 1.
GENERAL PROVISIONS
Article 1. Scope of regulation
This Circular provides for announcement of standard conformity and announcement of technical-regulation conformity and method to assess the conformity with standards and technical regulations.
Article 2. Subjects of application
This Circular applies to organizations, individuals and management agencies related to assessment of the conformity, announcement of standard conformity and announcement of technical-regulation conformity.
Article 3. Interpretation of terms
In this Circular, the following terms are construed as follows:
1. Announcement of standard conformity means self-announcement of organizations and individuals that products, goods, services, process, environment are conformable to respective standards.
2. Announcement of technical-regulation conformity means self-announcement of organizations and individuals that products, goods, services, process, environment are conformable to respective technical regulations.
3. The certifying organization conducting activities of certifying the standard conformity means organization already registered the certifying operation (hereinafter abbreviated to the certifying organization already registered) as prescribed in the Circular No. 08/2009/TT-BKHCN dated April 08, 2009, of the Minister of science and technology, guiding the requirements, orders of and procedures for registering operation of conformity assessment (hereinafter abbreviated to the Circular No. 08/2009/TT-BKHCN) and Circular No. 08/2009/TT-BKHCN) and Circular No. 10/2011/TT-BKHCN dated June 30, 2011, of the Minister of science and technology amending and supplementing a number of Articles of the Circular No. 08/2009/TT-BKHCN (hereinafter abbreviated to the Circular No. 10/2011/TT-BKHCN)
4. The certifying organization conducting activities of certifying the technical-regulation conformity means organization already registered as prescribed in Clause 3 of this Article and appointed by competent agencies to conduct activities of certifying the technical-regulation conformity (hereinafter abbreviated to the appointed certifying organizations).
5. The testing organization conducting activities of testing quality of products and goods means organization already registered the testing operation (hereinafter abbreviated to the testing organization already registered) as prescribed in the Circular No. 08/2009/TT-BKHCN and Circular No. 10/2011/TT-BKHCN
Article 4. Seal of standard conformity and seal of technical-regulation conformity
1. Seal of standard conformity and use of standard conformity seal
The registered certifying organization may prescribe the standard conformity seal including form, structure, and expression and use the standard conformity seal to issue to objects certified standard conformity and must satisfy the following basic requirements:
a) It must clear, not confuse with other seals;
b) It must express full signs of respective standard used as basis for certifying the standard conformity.
In case where organizations or individuals announce the standard conformity on the basis of results assessed by them, it not required to provide for form, structure, expression and the standard conformity seal is not allowed using.
2. Seal of technical-regulation conformity and use of technical-regulation conformity seal
a) The technical-regulation conformity seal has form, size as prescribed in Annex I of this Circular;
b) The technical-regulation conformity seal is used directly on products, goods or packages or in technical documents or on labels stuck on products, goods at apparent and readable positions;
c) The technical-regulation conformity seal must be ensured to not be erased easily and not be able to take it off then stick again;
d) The technical-regulation conformity seal may be enlarged or shrunk but the basic proportion and size of the technical-regulation conformity seal must be complied with provision in Annex I of this Circular and may be recognizable with naked eyes;
dd) The technical-regulation conformity seal must be designed and expressed in a same color and recognizable.
Article 5. Methods to assess the conformity
1. The conformity assessment is conducted according to one of the following methods:
a) Method 1: test on typical sample;
b) Method 2: test on typical sample and assess the manufacture process; supervise through testing on sample collected from market;
c) Method 3: test on typical sample and assess the manufacture process; supervise through testing on sample collected from manufacture area in association with assessment of the manufacture process;
d) Method 4: test on typical sample and assess the manufacture process; supervise through testing on sample collected from manufacture area and the market in association with assessment of the manufacture process;
dd) Method 5: test on typical sample and assess the manufacture process; supervise through testing on sample collected from manufacture area or the market in association with assessment of the manufacture process;
e) Method 6: assess and supervise the management system;
g) Method 7: test on and assess a batch of products, goods
h) Method 8: test or verify all products, goods
2. Content, orders and principle of using the methods to assess the conformity are prescribed in the Annex II of this Circular.
Article 6. Applying methods to assess the conformity
1. Methods to assess the standard conformity area applied to each kind of specific product, goods, service, the process, environment which are selected by organizations certifying the standard conformity or organizations, individuals announcing the standard conformity according to the methods to assess the conformity specified in Article 5 of this Circular. The selected method to assess the conformity must be suitable to the assessed object so as to ensure the reliability of result of assessing the conformity.
2.
The methods to assess the technical-regulation conformity applicable to specific products, goods, services, process, and environment are prescribed in the respective technical regulations
3. The methods to assess the conformity must be specified on certificate of the technical-regulation conformity.
Chapter 2.
ANNOUNCEMENT OF STANDARD CONFORMITY
Article 7. Principles for announcement of standard conformity
1. Objects of announcement of standard conformity include products, goods, services, process, environment specified in the respective standards. Announcement of standard conformity is a voluntary operation.
2. Announcement of conformity with respective standard is based on:
a) Result of certifying the standard conformity which is performed by the registered certifying organization or:
b) Result of self-assessing the conformity of organizations, individuals conducting announcement of standard conformity.
The test in serve for assessing the standard conformity must be conducted at the registered testing organizations.
Article 8. Orders of announcement of standard conformity
Announcement of standard conformity is conducted according to the following steps:
1. Step 1: Assessing the object conformity of announcement of standard conformity with the respective standard (hereinafter abbreviated to assessing the standard conformity).
a) The registered certifying organizations (third party) or organizations, individuals announcing the standard conformity (first party) shall assess the standard conformity.
The assessment of conformity shall comply with methods of assessing the conformity specified in clause 1 article 6 of this Circular;
b) Result of assessing the standard conformity specified in point a Clause 1 of this Article is basis for organizations and individuals to conduct announcement of standard conformity.
2. Step 2: Dossiers of announcing the standard conformity are registered at the Sub-Departments for Standards, Metrology and Quality in provinces or central-affiliated cities where production organizations or individuals register their enterprises or business households (hereinafter abbreviated to Sub-Departments).
Article 9. Dossier of register for announcement of standard conformity
Organizations and individuals announcing the standard conformity shall make 02 (two) sets of dossier of announcing the standard conformity, in which 01 (one) set of dossier shall be submitted directly or through post office to the Sub-Departments and 01 (one) set of dossier shall be kept by organizations and individuals. A prescribed dossier includes documents as follows:
1. In case of announcing the standard conformity based on result of certifying the standard conformity of the registered certifying organizations (the third party), dossier of announcing the standard conformity includes:
a) Announcement of standard conformity (made according to Form 2. CBHC/HQ specified in Annex III of this Circular);
b) Copies of originals of documentary evidences about production and business of organizations, individuals conducting announcement of standard conformity (written enterprise registration or the Business registration certificate or Business household registration certificate or investment certificate or decision on establishment or other papers as prescribed by law);
c) Copy of original of the standard used as basis for announcement;
d) Copy of original of the standard conformity certificate issued by the registered certifying organizations, enclosed with standard conformity seal specimen.
In the course of considering dossiers, if it is necessary, the copies may be considered or compared with the originals or the authenticated copies may be required for supplementation.
2. In case of announcing the standard conformity based on result of self assessment of the production and business organization or individual (the first party), dossier of announcing the standard conformity includes:
a) Announcement of standard conformity (made according to Form 2. CBHC/HQ specified in Annex III of this Circular);
b) Copies of originals of documentary evidences about production and business of organizations, individuals conducting announcement of standard conformity (written enterprise registration or the Business registration certificate or Business household registration certificate or investment certificate or decision on establishment or other papers as prescribed by law);
c) Copy of original of the standard used as basis for announcement;
d) In case where organizations, individuals announcing standard conformity have not yet been issued certificate of standard conformity about management system (ISO 9001, ISO 22000, HACCP...) by the registered certifying organizations, dossiers of announcing the standard conformity of organizations and individuals must have the production process enclosed with plan on quality control formulated and applied (according to form 1. KHKSCL specified in Annex III of this Circular) and plan on supervising the management system;
dd) In case where organizations, individuals announcing standard conformity have been issued certificate of standard conformity about management system (ISO 9001, ISO 22000, HACCP...) by the registered certifying organizations, dossiers of announcing the standard conformity of organizations and individuals must have copy of valid original of certificate of conforming standards about management system;
e) Report on assessing the standard conformity (made according to form 5. BCDG specified in Annex III of this Circular), enclosed with copy of original of the test result of sample within 12 months calculated to time of submitting dossier of announcing the standard conformity made by the registered testing organizations.
In the course of considering dossiers, if it is necessary, the copies may be considered or compared with the originals or the authenticated copies may be required for supplementation.
Article 10. Handling of dossier of announcing the standard conformity
Dossier of announcing the standard conformity send to the Sub-Department is handled as follows:
1. If dossiers of announcing the standard conformity fail to be sufficient as prescribed in Article 9 of this Circular, within 03 (three) working days after receiving dossier of announcing the standard conformity, Sub-Departments shall notify in writing to request for supplementation of kinds of papers as prescribed in Article 9 of this Circular to organizations and individuals announcing the standard conformity. Within 15 (fifteen) working days after Sub-Departments send the written request, if dossiers of announcing the standard conformity are not supplemented fully as prescribed, the Sub-Departments have right to cancel the handling of such dossiers.
2. If dossiers of announcing the standard conformity are sufficient as prescribed in Article 9 of this Circular, within 05 (five) working days after receiving dossier of announcing the standard conformity, Sub-Departments shall examine the validity of dossiers of announcing the standard conformity to solve as follows:
a) If dossier of announcing the standard conformity is sufficient and valid, the Sub-Department shall issue a notice of receiving dossier of announcing the standard conformity to organization or individual conducting announcement of standard conformity (made according to Form 3. TBTNHS specified in Annex III of this Circular). The notice of receiving dossier of announcing the standard conformity is valid according to value of certificate of standard conformity which is issued by the registered certifying organizations or it will be valid in 03 (three) years after leader of organization or individual has signed for certifying the report of assessing the standard conformity (in case where organization or individual self assess the standard conformity).
b) If dossier of announcing the standard conformity is sufficient but invalid, the Sub-Department shall issue a written notice to organization or individual conducting announcement of standard conformity about reason of refusal for receiving dossier.
Article 11. Responsibilities of organizations and individuals conducting announcement of standard conformity
1. To select methods to assess the conformity in suitability with object of announcing the standard conformity so as to ensure the reliability of assessment result.
2. To maintain continuously and take responsibility for the conformity of products, goods, services, process and environment as registered for announcement of standard conformity; maintain the quality control, testing and supervision on a periodical basis at the production and business facilities of organizations, individuals.
3. When detecting the non-conformity of products, goods, services, process, and environment already announced the standard conformity, during the circulation and use, organizations and individuals must:
a) Temporarily stop ex-warehousing and conduct recall of unconformable products and goods circulating on the market in case such unconformable products and goods have high risks of causing danger for users; stop the operation, exploitation of relevant process, services and environment as necessary;
b) To conduct measures to overcome the non-conformity;
c) To inform in writing to Sub-Departments about result of overcoming the non-conformity before continue putting the products, goods, services, process, environment into use, circulation, exploitation, and business.
4. To make and store dossiers of announcing the standard conformity as follows:
a) In case of announcing the standard conformity based on result of certifying the standard conformity of the registered certifying organizations (the third party), a stored dossier of announcing the standard conformity includes originals, copies of papers as prescribed in Clause 1 Article 6 and Dossier of assessment and supervision of the registered certifying organizations;
b) In case of announcing the standard conformity based on result of self-assessment of the production and business organizations and individuals (the first party), a stored dossier of announcing the standard conformity includes originals, copies of papers as prescribed in Clause 2 Article 9 and Dossier of self-assessment and supervision of organization or individual under the supervision plan.
5. To provide documents to prove assurance of conformity of products, goods, services, process and environment with the respective standard as required by competent state agencies.
6. To provide copy of original of notice on receiving dossier of announcing the standard conformity to the organization or individual business in products, goods, services, process and environment.
7. To implement the re-announcement when there is any change about content of the registered dossier of announcing the standard conformity or any change about feature, utility, characteristic of products, goods, or services already announced standard conformity.
Chapter 3.
ANNOUNCEMENT OF TECHNICAL-REGULATION CONFORMITY
Article 12. Principles for announcement of technical-regulation conformity
1. Objects of announcement of technical-regulation conformity include products, goods, services, process, environment specified in the national technical regulations promulgated by Ministries managing sectors and fields or specified in the local technical regulations promulgated by People’s Committees of provinces and central-affiliated cities. Announcement of technical-regulation conformity is a compulsory operation.
2. Announcement of conformity with technical regulations is based on one of two following cases:
a) Result of certifying the technical-regulation conformity as prescribed in the respective technical regulations and performed by the appointed certifying organizations;
b) Result of self-assessing the conformity of organizations, individuals conducting announcement of technical-regulation conformity.
The test in serve for assessing the technical-regulation conformity must be conducted at the registered testing organizations.
3. In case where products and goods is managed by many different technical regulations, organizations and individuals must register the announcement of technical-regulation conformity at respective specialized agencies and only permitted to use seal of technical-regulation conformity when such products and goods have been implemented fully measures of management as prescribed in the respective technical regulations.
Article 13. Orders of announcement of technical-regulation conformity
Announcement of technical-regulation conformity is conducted according to the following steps:
1. Step 1: Assessing the object conformity of announcement of technical-regulation conformity with the respective technical regulations (hereinafter abbreviated to assessing the technical-regulation conformity).
a) The registered certifying organizations (third party) or organizations, individuals announcing the technical-regulation conformity (first party) shall assess the technical-regulation conformity.
The assessment of technical-regulation conformity shall comply with methods of assessing the conformity specified in the respective technical regulations.
In case of using result of assessing the conformity made by a foreign organization of assessing the conformity, the foreign organization of assessing the conformity must be recognized as prescribed by law or appointed by competent state management agencies;
b) Result of assessing the technical-regulation conformity will be basis for organizations and individuals conducting announcement of technical-regulation conformity.
2. Step 2: Register the announcement of technical-regulation conformity at specialized agencies which are appointed by Ministries managing sectors, fields and People’s Committees of provinces and central-affiliated cities (hereinafter abbreviated to the specialized agencies).
Article 14. Dossier of register for announcement of technical-regulation conformity
Organizations and individuals announcing the technical-regulation conformity shall make 02 (two) sets of dossier of announcing the technical-regulation conformity, in which 01 (one) set of dossier shall be submitted directly or through post office to the specialized agencies and 01 (one) set of dossier shall be kept by organizations and individuals. A prescribed dossier includes documents as follows:
1. In case of announcing the technical-regulation conformity based on result of certifying the technical-regulation conformity of the appointed certifying organizations (the third party), dossier of announcing the technical-regulation conformity includes:
a) Announcement of technical-regulation conformity (made according to Form 2. CBHC/HQ specified in Annex III of this Circular);
b) Copies of originals of documentary evidences about production and business of organizations, individuals conducting announcement of technical-regulation conformity (written enterprise registration or the Business registration certificate or Business household registration certificate or investment certificate or decision on establishment or other papers as prescribed by law);
c) Copy of original of certificate of conforming to the respective technical regulations issued by the appointed certifying organization, enclosed with seal specimen of technical-regulation conformity which is issued to organization or individual by the appointed certifying organizations;
In the course of considering dossiers, if it is necessary, the copies may be considered or compared with the originals or the notarized copies may be required for supplementation;
2. In case of announcing the standard conformity based on result of self assessment of the production and business organization or individual (the first party), dossier of announcing the technical-regulation conformity includes:
a) Announcement of technical-regulation conformity (made according to Form 2. CBHC/HQ specified in Annex III of this Circular);
b) Copies of originals of documentary evidences about production and business of organizations, individuals conducting announcement of technical-regulation conformity (written enterprise registration or the Business registration certificate or Business household registration certificate or investment certificate or decision on establishment or other papers as prescribed by law);
c) In case where organizations, individuals announcing the technical-regulation conformity have not yet been issued certificate of conforming with standards about management system (ISO 9001, ISO 22000, HACCP...) by the registered certifying organizations, dossiers of announcing the technical-regulation conformity of organizations and individuals must have the production process enclosed with plan on quality control formulated and applied (according to form 1. KHKSCL specified in Annex III of this Circular) and plan on supervising the management system;
d) In case where organizations, individuals announcing technical-regulation conformity have been issued certificate of conforming with standards about management system (ISO 9001, ISO 22000, HACCP...) by the registered certifying organizations, dossiers of announcing the technical-regulation conformity of organizations and individuals must have copy of valid original of certificate of conforming standards about management system;
dd) Copy of original of the test result of sample within 12 months calculated to time of submitting dossier of announcing the technical-regulation conformity made by the registered testing organizations;
e) Report on assessing the technical-regulation conformity (made according to Form 5. BCDG specified in Annex III of this Circular) enclosed with seal specimen of technical-regulation conformity and relevant documents;
In the course of considering dossiers, if it is necessary, the copies may be considered or compared with the originals or the notarized copies may be required for supplementation.
Article 15. Handling of dossier of announcing the technical-regulation conformity
Dossier of announcing the technical-regulation conformity send to the Sub-Department is handled as follows:
1. If dossiers of announcing the technical-regulation conformity fail to be sufficient as prescribed in Article 14 of this Circular, within 03 (three) working days after receiving dossier of announcing the standard conformity, the specialized agencies shall notify in writing to request for supplementation of kinds of papers as prescribed to organizations and individuals announcing the technical-regulation conformity. Within 15 (fifteen) working days after the specialized agencies send the written request, if dossiers of announcing the technical-regulation conformity are not supplemented fully as prescribed, the specialized agencies have right to cancel the handling of such dossiers.
2. If dossiers of announcing the technical-regulation conformity are sufficient as prescribed in Article 14 of this Circular, within 05 (five) working days after receiving dossier of announcing the technical-regulation conformity, the specialized agencies shall examine the validity of dossiers of announcing the standard conformity:
a) If dossier of announcing the standard conformity is sufficient and valid, the specialized agencies shall issue a notice of receiving dossier of announcing the technical-regulation conformity to organization or individual conducting announcement of technical-regulation conformity (made according to Form 3. TBTNHS specified in Annex III of this Circular).
The notice of receiving dossier of announcing the technical-regulation conformity is valid according to value of certificate of technical-regulation conformity which is issued by the appointed certifying organizations or it will be valid in 03 (three) years after leader of organization or individual has signed for certifying the report of assessing the technical-regulation conformity (in case where organization or individual self assess the technical-regulation conformity);
b) If dossier of announcing the technical-regulation conformity is sufficient but invalid, the specialized agencies shall issue a written notice to organization or individual conducting announcement of technical-regulation conformity about reason of refusal for receiving dossier.
Article 16. Responsibilities of organizations and individuals conducting announcement of technical-regulation conformity
1. To inform on suitable media about their announcement of technical-regulation conformity to ensure that persons using such products and goods can access to information easily.
2. To maintain continuously and take responsibility for the conformity of products, goods, services, process and environment which have been announced technical-regulation conformity; maintain the quality control, testing and supervision on a periodical basis.
3. To use seal of technical-regulation conformity for products and goods which have been announced technical-regulation conformity as prescribed in clause 2 Article 4 of this Circular before putting them into circulation on market. To make logbook and annually report use of seal of technical-regulation conformity to the appointed certifying organizations.
4. When detecting the non-conformity of products, goods, services, process, and environment already announced the technical-regulation conformity, during the circulation and use, organizations and individuals must:
a) Timely inform in writing about the non-conformity to the specialized agencies;
b) Temporarily stop ex-warehousing and conduct recall of unconformable products and goods circulating on the market in case such unconformable products and goods have high risks of causing danger for users; stop the operation, exploitation of relevant process, services and environment as necessary;
c) To conduct measures to overcome the non-conformity;
d) To inform in writing to the specialized agencies about result of overcoming the non-conformity before continue putting the products, goods, services, process, environment into use, circulation, exploitation, and business.
5. To make and store dossiers of announcing the technical-regulation conformity to do as basis for examination, inspection of the state management agencies as follows:
a) In case of announcing the technical-regulation conformity based on result of certifying the technical-regulation conformity of the appointed certifying organizations (the third party), a stored dossier of announcing the technical-regulation conformity includes originals, copies of papers as prescribed in Clause 1 Article 14 and Dossier of assessment and supervision of the appointed certifying organizations;
b) In case of announcing the technical-regulation conformity based on result of self-assessment of the production and business organizations and individuals (the first party), a stored dossier of announcing the technical-regulation conformity includes originals, copies of papers as prescribed in Clause 2 Article 14 and Dossier of self-assessment and supervision of organization or individual under the supervision plan.
6. To provide documents to prove assurance of conformity of products, goods, services, process and environment with the respective technical regulations as required by competent state agencies.
7. To provide copy of original of certificate of technical-regulation conformity, notice on receiving dossier of announcing the technical-regulation conformity to the organization or individual business in products, goods, services, process and environment.
8. To implement the re-announcement when there is any change about content of the registered dossier of announcing the technical-regulation conformity or any change about feature, utility, characteristic of products, goods, or services already announced technical-regulation conformity.
Chapter 4.
ORGANIZATION OF IMPLEMENTATION
Article 17. Responsibilities of management agencies
1. Responsibilities of Ministries managing sectors and fields, and People’s Committees of provinces, central-affiliated cities:
a) To direct activities involving announcement of technical-regulation conformity as prescribed in this Circular when promulgating respective technical regulations for management;
b) To direct focal agencies in charge of managing activities in announcement of technical-regulation conformity in the assigned fields; notify list of focal agencies for the relevant organizations and individuals for implementation and send list to the Ministry of science and technology for coordination and management;
c) To assign duty for receiving dossier of announcing the technical-regulation conformity to the specialized agencies;
d) Annually, to sum up situation of appointing the organizations for assessing the conformity, notify the Ministry of Science and Technology for coordination in management; or irregularly, as required, to sum up and report on situation of announcing the technical-regulation conformity to the Ministry of Science and Technology for summing up and reporting to the Prime Minister.
2. Responsibility of the Directorate for Standards, Metrology and Quality shall act as an appointed focal agency as prescribed in point b Clause 1 of this Article:
a) To help the Ministry of Science and Industry for unified management and guiding activities involving assessment of the conformity, announcement of standard conformity and announcement of technical-regulation conformity;
b) To coordinate with focal agencies at Central level under Ministries managing sectors and fields, the Departments of science and technology in provinces and central-affiliated cities, in urging, guiding implementation of announcement of standard conformity, announcement of technical-regulation conformity as prescribed in this Circular;
c) To monitor situation of announcement of standard conformity and announcement of technical-regulation conformity for products, goods, services, process, environment under management responsibility of Ministry of science and technology on the basis of reports of Sub-Departments of Standard, Metrology and Quality; to monitor the appointment of Ministries managing sectors, fields about activities of assessing the conformity.
3. Responsibilities of the appointed focal agencies, as prescribed in point b Clause 1 of this Article, and under Ministries managing sectors and fields, and People’s Committees of provinces, central-affiliated cities:
a) Monitoring and managing activities of registration for announcement of technical-regulation conformity conducted by the specialized agencies; coordinating with the Directorate for Standards, Metrology and Quality in managing activities of announcement of technical-regulation conformity; annually, summing up and reporting to send to Ministries managing sectors, fields and People’s Committees of provinces, central-affiliated cities, involving situation of appointment of assessing the conformity, concurrently sending them to the Directorate for Standards, Metrology and Quality for coordination in management;
b) Summing up situation of announcement of technical-regulation conformity conducted by the specialized agencies and annually, or irregularly as required, reporting to the relevant Ministries managing sectors, fields and People’s Committees of provinces and central-affiliated cities.
4. Responsibilities of the specialized agencies which are appointed by the Ministries managing sectors and fields, and People’s Committees of provinces, central-affiliated cities:
a) Receiving dossiers of registration and managing dossiers of announcement of technical-regulation conformity; cancelling, suspending results of receiving dossiers of announcing the technical-regulation conformity for products, goods, services, process, environment managed by the national technical regulations which are promulgated by Ministries managing sectors and fields and the local technical regulations related to fields under their management;
b) To post publicly on their websites about situations of announcement of technical-regulation conformity with the following contents:
- Name of organizations and individuals conducting announcement of technical-regulation conformity;
- Products and goods announced the technical-regulation conformity;
- Number of technical regulations;
- Assessment type: The first party (name of organizations and individuals) or third party (name of the appointed certifying organizations).
c) To coordinate with the Sub-Department of Standards, Metrology and Quality;
d) Annually or irregularly as required, to sum up and report to the focal agencies about list of products, goods, services, process and environment which have been registered for announcement of technical-regulation conformity (made according to Form 4. BCTNHS specified in Annex III of this Circular).
5. Responsibilities of Sub-Departments of Standards, Metrology and Quality, under Departments of science and technology in provinces and central-affiliated cities:
a) To receive registration and management of dossiers of announcement of standard conformity; cancel, suspend results of receiving dossier of announcement of standard conformity of business and production organizations and individuals in localities and post publicly on websites of Departments of science and technology or Sub-Departments of Standards, Metrology and Quality about situation of announcement of standard conformity;
b) To receive dossiers of registration and manage dossiers of announcement of technical-regulation conformity; cancel, suspend results of receiving dossiers of announcing the technical-regulation conformity for products, goods, services, process, environment managed by the national technical regulations which are promulgated by the Ministry of Science and Technology and the local technical regulations related to fields under their management; post publicly on websites of Departments of science and technology or Sub-Departments of Standards, Metrology and Quality on localities about situation of announcement of technical-regulation conformity with the following contents:
- Name of organizations and individuals conducting announcement of technical-regulation conformity;
- Products and goods announced the technical-regulation conformity;
- Number of technical regulations;
- Assessment type: The first party (name of organizations and individuals) or third party (name of the appointed certifying organizations).
c) To coordinate with the specialized agencies in localities in providing information about announcement of standard conformity to facilitate for examination on quality of products and goods;
d) Annually or irregularly as required, to sum up and report to the Directorate for Standards, Metrology and Quality about situation of receiving dossiers of announcement of standard conformity, announcement of technical-regulation conformity (made according to Form 4. BCTNHS specified in Annex III of this Circular) as prescribed in point a, b this Clause.
Article 18. Examination, inspection and handling of violations
1. The competent state management agencies shall conduct examination, inspection and handling of violations in announcement of standard conformity and announcement of technical-regulation conformity as prescribed in this Circular and other relevant current regulations.
2. Organizations and individuals infringing regulations on announcement of standard conformity and announcement of technical-regulation conformity, depend on nature and seriousness of violations shall be handled as prescribed by relevant current law.
Article 19. Provisions of implementation
This Circular takes effect on January 27, 2013, and replaces the Decision No. 24/2007/QD-BKHCN dated September 28, 2007, of the Minister of science and technology, on promulgating regulation on certifying standard conformity, certifying technical-regulation conformity and announcement of standard conformity, announcement of technical-regulation conformity.
Article 20. Organization of implementation
1. The Ministers, Heads of ministerial-level agencies, Chairpersons of People’s Committees of provinces and central-affiliated cities shall carry out implementation of this Circular.
2. The General Director of Directorate for Standards, Metrology and Quality shall guide and organize implementation of this Circular.
3. In the course of implementation, any arising problems should be reported timely in writing to the Ministry of science and technology for research, amendment and supplementation.
|
|
FOR
THE MINISTER OF SCIENCE AND TECHNOLOGY |
ANNEX I
SHAPE AND SIZE OF SEAL OF TECHNICAL-REGULATION CONFORMITY (Enclosed with the Circular No. 28/2012/TT-BKHCN dated December 12, 2012, of the Minister of Science and Technology)
SHAPE AND SIZE OF SEAL OF TECHNICAL-REGULATION CONFORMITY
1. Seal of technical-regulation conformity has the shape as described at picture 1.

Picture 1. Shape of seal of technical-regulation conformity
2. The basic size for designing seal of technical-regulation conformity specified in picture 2.
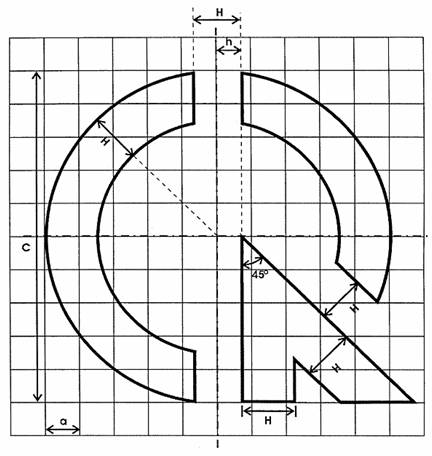
Picture 2. The basic size of seal of technical-regulation conformity
Notes:
H = 1.5 a
h = 0.5 H
C = 7.5 H
ANNEX II
CONTENT, ORDER AND PRINCIPLE OF USING METHODS TO ASSESS THE CONFORMITY (Enclosed with the Circular No. 28/2012/TT-BKHCN dated December 12, 2012, of the Minister of Science and Technology)
CONTENT, ORDERS AND PRINCIPLE OF USING THE METHODS TO ASSESS THE CONFORMITY
I. Method 1: test on typical sample
Method 1 by test on typical sample of products and goods to conclude about the conformity. Conclusion about the conformity is valid for model, type of products and goods which are taken sample for test.
1. Content and order of implementing the main activities in Method 1 include:
Taking sample:
Taking typical samples for products and goods. Typical sample of products and goods is sample representing for a specific model or type of products or goods produced according to a same design, in a same condition and made of same raw materials.
Quantity of samples must be full for testing and storing sample.
1.2. Assessing the conformity of the testing sample:
Samples of products and goods are conducted test at laboratory already registered for operational sector of testing as prescribed by law, may include laboratory of producers. To prioritize for using of laboratory already appointed and recognized.
Characteristics of products, goods for testing and method of testing shall be stipulated in the respective standards and technical regulations.
1.3. Handling of results of assessing the conformity
To consider characteristics of products, goods through result of testing samples in comparison with requirements of the respective standards and technical regulations.
1.4. Conclusion about the conformity
To conclude about the conformity of products and goods in comparison with requirements of the respective standards and technical regulations. Products and goods will be considered as conformable if all criteria of the testing sample are conformable with the prescribed level of the respective standards and technical regulations.
2. Principle of using the method 1
Method 1 is used to assess the conformity of products and goods with the following conditions:
a) Design of products and goods allow defining clearly products and goods according to each typical model, type;
b) Fail to consider requirements to ensure maintenance of quality stability.
II. Method 2: Test on typical samples and assessment on the production process; supervision through testing samples taken on the market
Method 2 bases on result of test on typical samples and assessment of the manufacture process to conclude about the conformity of products and goods. Assessment of supervision is conducted after that and conducted through test on samples of products and goods taken on the market.
1. Content and order of implementing the main activities in Method 2 include:
Taking sample:
Conducting as prescribed in item 1.1 of method 1.
1.2. Assessing the conformity of the testing sample:
Conducting as prescribed in item 1.2 of method 1.
1.3. Assessing the conformity of the production process:
Assessment of production process must consider full conditions of control of producers related to forming products with the aim to ensure maintenance of stability of quality of products and goods. Conditions for control include:
a) Control of technical dossiers of products (design documents, technical standards of products);
b) Control of all production process from input, through intermediate stages, till as finishing products included process of packing, loading and unloading, storage in warehouses and transport of products;
c) Control of quality of raw materials, semi-products and finished products;
d) Control of technological equipment and equipment for metrology, examination and testing;
dd) Control of professional skill qualification of workers and technical officers;
e) Other necessary technical contents.
In case where producers have gotten certificate of quality control system of the certifying organizations registered in sector of certifying or recognized for sector of producing products and goods which are assessed, It is not required to assess the process of production. However, if there is proof about failing to maintain validity of quality control system, the certifying organizations should conduct assessment of the production process, concurrently report to the Directorate for Standards, Metrology and Quality.
1.4. Handling of results of assessing the conformity:
To consider characteristics of products, goods through result of testing samples in comparison with requirements of the respective standards and technical regulations.
To consider the conformity of the production process in comparison with requirements specified in item 1.3 of this method.
1.5. Conclusion about the conformity
To conclude about the conformity of products and goods in comparison with requirements of the respective standards and technical regulations. Products and goods shall be considered as conformable if they ensure full two following conditions:
a) All criteria of the testing sample are conformable to the prescribed level of the respective standards and technical regulations.
b) Result of assessing the production process is conformable to requirement.
Conclusion about the conformity of products and goods will be valid maximally in 3 years, and provided that such products and goods are assessed supervision.
1.6. Supervision:
During valid time of conclusion about the conformity, products and goods must be assessed and supervised through test of samples taken on the market. Frequency of assessment and supervision must ensure not exceeding 12 months once.
The test of samples of products and goods is conducted as prescribed in items 1.1, 1.2 and 1.3 of method 1.
Result of assessing supervision will be used as basis for deciding on maintenance, suspension or cancellation of conclusion about the conformity.
2. Principle of using the method 2
Method 2 is used to assess the conformity of products and goods with the following conditions:
a) Products and goods under subject of having risks involving safety, health and environment at low level.
b) Design of products and goods allow defining clearly products and goods according to each typical model, type;
c) The maintenance of stability and quality characteristics of products and goods should be paid attention during the production;
d) Quality of products and goods able to be changed during course of distribution and circulation on the market;
dd) Organizations and individuals producing and trading in products and goods have effective measures to recall products and goods from the market when detecting unconformable products and goods during supervision.
III. Method 3: test on typical samples and assess the manufacture process; supervise through testing on samples collected from manufacture area in association with assessment of the manufacture process
Method 3 bases on result of test on typical samples and assessment of production process to conclude about the conformity. Assessment of supervision will be conducted through test of samples of products and goods taken from manufacture area in association with assessment of the manufacture process.
1. Content and order of implementing the basic activities in Method 3 include:
Taking sample:
Conducting as prescribed in item 1.1 of method 1.
1.2. Assessing the conformity of the testing sample:
Conducting as prescribed in item 1.2 of method 1.
1.3. Assessing the conformity of the production process:
Conducting as prescribed in item 1.3 of method 2.
1.4. Handling of results of assessing the conformity:
Conducting as prescribed in item 1.4 of method 2.
1.5. Conclusion about the conformity
Conducting as prescribed in item 1.5 of method 2.
1.6. Supervision:
During valid time of conclusion about the conformity, products and goods must be assessed and supervised through test of samples taken at the production area in association with assessment of the production process. Frequency of assessment and supervision must ensure not exceeding 12 months once.
The test of samples of products and goods is conducted as prescribed in items 1.1, 1.2 and 1.3 of method 1.
The assessment of the production process is conducted as prescribed in items 1.3 of method 2.
Result of assessing supervision will be used as basis for deciding on maintenance, suspension or cancellation of conclusion about the conformity.
2. Principle of using the method 3
Method 3 is used to assess the conformity of products and goods with the following conditions:
a) Products and goods under subject of having risks causing loss of safety, health and environment at a level higher than products and goods assessed under the method 2;
b) Design of products and goods allow defining clearly products and goods according to each typical model, type;
c) The maintenance of stability and quality characteristics of products and goods should be paid attention during the production;
d) Quality of products and goods, on nature, is not changed or changed a little during distribution and circulation on the market;
dd) It is difficult to have effective measures to recall products and goods from the market when detecting unconformable products and goods during supervision.
IV. Method 4: test on typical sample and assess the manufacture process; supervise through testing on sample collected from manufacture area and the market in association with assessment of the manufacture process
Method 4 bases on result of test on typical samples and assessment of production process to conclude about the conformity. Assessment of supervision after that will be conducted through test of samples of products and goods taken from manufacture area and market in association with assessment of the manufacture process.
1. Content and order of implementing the basic activities in the Method include:
Taking sample:
Conducting as prescribed in item 1.1 of Method 1.
1.2. Assessing the conformity of the testing samples:
Conducting as prescribed in item 1.2 of Method 1.
1.3. Assessing the conformity of the production process:
Conducting as prescribed in item 1.3 of Method 2.
1.4. Handling of results of assessing the conformity:
Conducting as prescribed in item 1.4 of Method 2.
1.5. Conclusion about the conformity
Conducting as prescribed in item 1.5 of Method 2.
1.6. Supervision:
During valid time of notice about the conformity, products and goods must be assessed and supervised through test of samples taken at the production area and market in association with assessment of the production process. Frequency of assessment and supervision must ensure not exceeding 12 months once.
The test of samples of products and goods is conducted as prescribed in items 1.1, 1.2 and 1.3 of method 1.
The assessment of the production process is conducted as prescribed in items 1.3 of method 2.
Result of assessing supervision will be used as basis for deciding on maintenance, suspension or cancellation of conclusion about the conformity.
2. Principle of using the method 4:
Method 4 is used to assess the conformity of products and goods with the following conditions:
a) Products and goods under subject of having risks causing loss of safety, health and environment at a level higher than products and goods assessed under the method 3;
b) Design of products and goods allow defining clearly products and goods according to each typical model, type;
c) The maintenance of stability and quality characteristics of products and goods should be paid attention during the production;
d) Quality of products and goods is able to lose stability during production, and able to be changed during distribution and circulation on the market;
dd) There are measures allowing recall of products and goods from the market when detecting unconformable products and goods during supervision.
V. Method 5: test on typical sample and assess the manufacture process; supervise through testing on sample collected from manufacture area or the market in association with assessment of the manufacture process.
Method 5 based on result of test on typical samples and assessment of production process to conclude about the conformity. The assessment of supervision is conducted through test on samples taken at production area or on market in association with assessment of the manufacture process.
1. Content and order of implementing the basic activities in Method 5 include:
Taking sample:
Conducting as prescribed in item 1.1 of Method 1.
1.2. Assessing the conformity of the testing samples:
Conducting as prescribed in item 1.2 of Method 1.
1.3. Assessing the conformity of the production process:
Conducting as prescribed in item 1.3 of Method 2.
1.4. Handling of results of assessing the conformity:
Conducting as prescribed in item 1.4 of Method 2.
1.5. Conclusion about the conformity
Conducting as prescribed in item 1.5 of Method 2.
1.6. Supervision:
During valid time of conclusion about the conformity, products and goods must be assessed and supervised through test of samples taken at the production area or on market in association with assessment of the production process. Frequency of assessment and supervision must ensure not exceeding 12 months once.
The test of samples of products and goods is conducted as prescribed in items 1.1, 1.2 and 1.3 of method 1.
The assessment of the production process is conducted as prescribed in items 1.3 of method 2.
Result of assessing supervision will be used as basis for deciding on maintenance, suspension or cancellation of notice about the conformity.
2. Principle of using the method 5:
Method 5 is used to assess the conformity of products and goods with the following conditions:
a) It is necessary to use a method with high reliability as method 4, but it is allowed to be flexible in using supervision measures to cut down costs;
b) It is necessary to use a method which is applied popularly aiming to direct to mutual recognition of results of assessing the conformity.
VI. Method 6: Assessing and supervising the management system
Method 6 bases on assessment of the management system to conclude about the conformity of management system with the respective standards and technical regulations.
1. Content and order of implementing the main activities in Method 6 include:
1.1. Assessing the conformity of management system:
- The management system is assessed under the respective standards and technical regulations.
- Report on result of assessment in comparison with the respective standards and technical regulations.
1.2. Conclusion about the conformity
Base on result of assessment, conclusion about the conformity of management system with the respective standards and technical regulations.
Conclusion about the conformity of management system which is valid maximally 3 years, and provided that management system is assessed and supervised.
1.3. Supervision of management system.
- Supervision through assessment of management system with frequency of assessment and supervision must ensure not exceeding 12 months once.
- Result of assessment is basis for deciding on further maintenance, suspension, cancellation of the conformity of management system.
2. Principle of using the method 6:
Method 6 is used to assess the conformity of process, service, environment with management system under the respective standards and technical regulations.
VII. Method 7: test on and assess a batch of products, goods
The method 7 bases on result of test on samples of products and goods which are taken according to method of statistical probability for a batch of products and goods to give out conclusion about the conformity of batches. Conclusion about the conformity will be valid for the specific batch of products and goods, and it is not required to conduct the following supervision measures.
1. Content and order of implementing the main activities in Method 7 include:
Taking sample:
The test sample is sample taken under method of statistical probability, ensures the representative nature for all batch.
Quantity of samples must be full for testing and storing sample.
1.2. Assessing the conformity of the testing samples:
Conducting as prescribed in item 1.2 of Method 1.
1.3. Handling of results of assessing the conformity:
To consider characteristics of products, goods through result of testing samples in comparison with the respective standards and technical regulations.
1.4. Conclusion about the conformity:
The batch of products and goods will be considered as conformable if quantity of the testing samples with result of non-conformity is in the allowed limitation.
The batch of products and goods will be considered as unconformable with regulations if quantity of the testing samples with result of non-conformity exceeds the allowed limitation.
2. Principle of using the method 7:
The method 7 is used to assess the conformity of products and goods with the following conditions:
a) Products and goods are divided under identical batches;
b) Fail to consider requirements to ensure maintenance of quality stability.
VIII. Method 8: test or verify all products, goods
Method 8 bases on result of testing or verifying all products, goods to conclude the conformity before putting into circulation and use. Conclusion about the conformity will be valid for each single product or goods, and it is not required to conduct the following supervision measures.
1. Content and order of implementing the main activities in Method 8 include:
1.1. Defining the products, goods which need to be tested or verified;
1.2. Assessing the conformity of products, goods:
a) Testing or verifying products and goods is conducted by laboratories, the verifying rooms which have been registered for the capable operational sector at production place, installment place, use place or laboratories, the verifying rooms.
To prioritize for using of laboratories, the verifying rooms already recognized.
b) Characteristics of products, goods which need to be tested, verified, and method of testing, verifying shall be stipulated in the respective standards and technical regulations.
1.3. Handling of results of assessing the conformity:
Consider characteristics of products and goods through result of testing or result of verifying in comparison with requirement.
1.4. Conclusion about the conformity:
Products and goods will be considered as conformable if all criteria of the tested or verified products and goods are conformable with the prescribed level of the respective standards and technical regulations.
2. Principle of using the method 8:
The method 8 is used to assess the conformity of products, goods with strict requirements on safety before putting into circulation and use.
ANNEX III
THE FORMS USED IN ANNOUNCEMENT OF STANDARD
CONFORMITY AND ANNOUNCMENT OF TECHNICAL-REGULATION CONFORMITY
(Enclosed with the Circular No. 28/2012/TT-BKHCN dated December 12, 2012, of
the Minister of Science and Technology)
1. Plan on quality control:
Form 1. KHKSCL
2. Announcement of standard conformity / announcement of technical-regulation conformity:
Form 2. CBHC/HQ
3. Notice of receiving dossier of announcement of standard conformity / announcement of technical-regulation conformity:
Form 3. TBTNHS
4. Report on situation of receiving dossiers of announcement of standard conformity / announcement of technical-regulation conformity:
Form 4. BCTNHS
5. Report on assessing the standard conformity and technical-regulation conformity:
Form 5. BCDS
Form
1. KHKSCL
28/2012/TT-BKHCN
PLAN ON QUALITY CONTROL
Products/goods/services/process/environment: ……………………………….
|
The specific production processes |
Plan on quality control |
||||||
|
Criteria of supervision and control |
Standards/ technical regulations |
Frequency of taking sample/ size of sample |
Equipment for testing/examination |
Methods of test/examination |
The recording table |
Note |
|
|
(1) |
(2) |
(3) |
(4) |
(5) |
(6) |
(7) |
(8) |
|
|
|
|
|
|
|
|
|
|
|
………….,
date........month........year ..…. |
Form
2. CBHC/HQ
28/2012/TT-BKHCN
|
SOCIALIST REPUBLIC OF
VIET NAM
ANNOUNCEMENT OF STANDARD CONFORMITY / ANNOUNCEMENT OF TECHNICAL-REGULATION CONFORMITY No. ………………………….. Name of organization or individual: ……… …………………………………………………………………………… Address: ……………………………………………………..……………………………………………… Telephone number: ………………………….. Facsimile: …………………………………………………………… E-mail: ……………………………………………………..……………………………………………… ANNOUNCING: Products, goods, process, services, environment (name, model, type, label, technical characteristic,… ) …………………………..………………………………………..………………………………………… ……………………………………………………………………..……………………………………….. conforming to standard/technical regulations (number, sign, name) …………………………..………………………………………..………………………………………… ……………………………………………………………………..……………………………………….. Added information (based on announcement of standard conformity or announcement of technical-regulation conformity, and method to assess the conformity. ……………………………………………………..………………………………………………………… ……………………………………………………..………………………………………………………… ……………………………………………………..………………………………………………………… .....(Name of organization or individual) …….. hereby commits and takes responsibility for the conformity of………. (products, goods, process, services, environment)… which are produced, traded, preserved, transported, used and exploited by it/him/her.
|
|
|
|
………….,
date........month........year …. (Signature, position, seal)
|
Form
3. TBTNHS
28/2012/TT-BKHCN
|
NAME
OF SUPERIOR MANAGEMENT AGENCY |
SOCIALIST REPUBLIC OF
VIET NAM |
|
No.: …….../TB-…… |
………, date........month........year ….. |
NOTICE
OF RECEIVING DOSSIER OF ANNOUNCEMENT OF STANDARD CONFORMITY / ANNOUNCEMENT OF TECHNICAL-REGULATION CONFORMITY
……. (Name of agency receiving announcement)……….. certifies that dossier of announcing the standard conformity/ the technical-regulation conformity No. ......... dated ........... of ............. (name of organization or individual)…………………
Address of organization or individual: …………………………………………………………………………………
has been received.
For products, goods, process, services, environment (name, model, type, label, technical characteristic,…): ……………………………………………………………………………………………….
conforming to standard (number, sign, name of standard) /technical regulations (number, sign, name of technical regulations) and be valid till date ……………….. (or state: Be valid in 3 years from date ……………..).
This notice records the commitment of organization or individual. This notice has no value to certify that products, goods, services, process, environment are conformable to the respective standards/ technical regulations.
(Name of organization or individual) …………must entirely be responsible for the conformity of products, goods, process, services, and environment, which are produced, traded, preserved, transported, used and exploited by it/him/her.
|
Receivers: |
Competent
representative of agency receiving the announcement |
Form
4. BCTNHS
28/2012/TT-BKHCN
|
NAME
OF SUPERIOR MANAGEMENT AGENCY |
SOCIALIST REPUBLIC OF
VIET NAM |
|
No.: …….../TB-…… |
………, date........month........year ….. |
REPORT
OF RECEIVING DOSSIER OF ANNOUNCEMENT OF STANDARD CONFORMITY / ANNOUNCEMENT OF TECHNICAL-REGULATION CONFORMITY
(From date ….. month …….. year ……… to date ….. month …….. year ………)
|
No. |
The receiving number |
Name of organization or individual conducting announcement |
Name of products, goods, process, services, environment |
Standards/ technical regulations |
Assessment type |
Note |
|
|
The first party (name of the certifying organization already registered / appointed) |
The third party (self assessment) |
|
|||||
|
1 |
|
|
|
|
|
|
|
|
2 |
|
|
|
|
|
|
|
|
.... |
|
|
|
|
|
|
|
Total dossiers of announcement of standard conformity / announcement of technical-regulation conformity already received:…………………………………………………………
|
Receivers: |
Competent
representative of agency receiving the announcement |
Form
5. BCDG
28/2012/TT-BKHCN
|
NAME
OF SUPERIOR MANAGEMENT AGENCY (if any) |
SOCIALIST REPUBLIC OF
VIET NAM |
|
No.: ……...…… |
………, date........month........year ….. |
REPORT
ON ASSESSING THE STANDARD CONFORMITY AND TECHNICAL-REGULATION CONFORMITY
1. Assessment date: ..............................................................................................................
2. Assessment place: ..............................................................................................................
3. Product name: ..............................................................................................................
4. Number of standards / technical regulations applied: .............................................................................
5. Name of organization testing the product: ................................................................
6. Assessment on result of testing according to the applied standards / technical regulations and the effectiveness of application, implementation of the production process: ............................................................................. .................................... ............................
...............................................................................................................................................
...............................................................................................................................................
...............................................................................................................................................
...............................................................................................................................................
...............................................................................................................................................
7. Other content (if any): .................................... .................................... .............................
8. Conclusion:
Product conforms to standard/technical regulations.
Product fails to conform to standard/technical regulations.
|
The assessing person (signature and full name) |
Certified
by leader of organization or individual |









