Nội dung toàn văn Decision 3638/QD-BYT 2021 introducing interim guidelines COVID19 monitoring and prevention
|
MINISTRY OF HEALTH |
SOCIALIST REPUBLIC OF VIETNAM |
|
No. 3638/QD-BYT |
Hanoi, July 30, 2021 |
DECISION
INTRODUCING INTERIM GUIDELINES FOR COVID-19 MONITORING AND PREVENTION
MINISTER OF HEALTH
Pursuant to the Government’s Decree No.75/2017/ND-CP dated June 20, 2017 on functions, duties, powers and organizational structure of the Ministry of Health;
At the request of General Director of General Department of Preventive Medicine, Ministry of Health,
HEREBY DECIDES:
Article 1. Promulgated together with this Decision are the interim guidelines for COVID-19 monitoring and prevention.
Article 2. Interim guidelines for COVID-19 monitoring and prevention shall be implemented in preventive healthcare establishments and healthcare establishments nationwide.
Article 3. This Decision takes effect from the date on which it is signed and supersedes Decision No. 3468/QD-BYT dated August 07, 2020 by the Ministry of Health introducing interim guidelines for COVID-19 monitoring and prevention.
Article 4. Head of Office of the Ministry of Health; Chief Inspector of the Ministry of Health; heads of affiliates of the Ministry of Health; heads of Institutes of Hygiene and Epidemiology; heads of Pasteur Institutes; directors of hospitals affiliated to the Ministry of Health; Directors of Departments of Health; heads of health units of other ministries; and heads of relevant units shall implement this Decision.
|
|
P.P. THE MINISTER |
INTERIM GUIDELINES
COVID-19 MONITORING AND PREVENTION
(Enclosed with Decision No. 3638/QD-BYT dated July 30, 2021 by Ministry of Health)
I. GENERAL CHARACTERISTICS
COVID-19 is a group-A acute infectious disease caused by the SARS-CoV-2 virus. This virus mutates regularly, resulting in more infectious variants. As of July 2021, Vietnam records 07 SARS-CoV-2 variants, including variants common in Europe, Africa, the UK and India; the outbreak starting from 27/4/2021 records the Delta (B. 1. 617. 2, first found in India) and Alpha (B. 1. 1. 7, first found in the UK) variants; the Delta variant is classified as a “variant of concern” by WHO and is 50% more infectious than the Alpha variant.
COVID-19 spreads from human to human via the respiratory route. Incubation period is approximately 14 days. Most COVID-19 patients (more than 60%) have no clinical manifestation. In patients with symptoms, clinical manifestations vary, ranging from fever, cough, throat pain, fatigue, body ache, reduction or loss of taste or smell, breathing difficulty to severe pneumonia, acute respiratory failure and death, especially in those with underlying conditions or chronic diseases and the elderly. Vaccines against COVID-19 are available but no effective cure has been found.
These interim guidelines are formulated and updated based on actual COVID-19 containment, provide for COVID-19 monitoring, prevention and control as appropriate to current situation and shall be implemented by provincial governments, health units and relevant units according to actual local situation.
II. MONITORING GUIDELINES
1. Definition of cases and contacts
1.1. Suspected cases (monitored cases)
Suspected cases are those with at least two of the following symptoms: fever, cough, throat pain, difficulty breathing, body ache - fatigue - chill; reduction or loss of taste or smell; or testing positive for SARS-CoV-2 via screening test.
1.2. Confirmed cases (F0 cases)
Confirmed cases are persons testing positive for SARS-CoV-2 via genetic material detection testing by testing facilities permitted to perform such testing by the Ministry of Health.
1.3. Close contacts (F1 cases)
F1 cases are persons having close contact within 2 meters or staying in an enclosed space in a workplace, factory, learning place, recreational place, etc. or in a section of a transport vehicle with an F0 case during the infectious period of the F0 case, specifically:
- For F0 cases with symptoms: from 3 days before symptom onset of the F0 case to the time where the F0 case is placed under medical isolation. Symptom onset shall be the day where the patient feels the first unusual health symptom(s) such as fatigue; lack of appetite; body ache; chill; reduction or loss of taste or smell; fever; cough; throat pain, etc.
- For asymptomatic F0 cases:
+ If the infection source of the F0 case has been identified: from the time where the F0 case first has contact with the infection source to the time where the F0 case is placed under medical isolation.
+ If the infection source of the F0 case has not been identified: from 14 days before the time where the sample testing positive for the disease is collected from the F0 case to the time where the F0 case is placed under medical isolation.
* Some common close contacts include:
- Persons living in the same household, house or room.
- Persons directly caring for or visiting a confirmed case or patients staying in the same room as a confirmed case.
- Persons working in the same team or office.
- Persons in a travel group, business team, friend group, party, meeting, classroom, religious gathering or club, on a transport vehicle, etc. having contact with a confirmed case.
1.4. Second-hand contacts (F2 cases)
F2 cases are persons having close contact within 2 meters with an F1 case from the time where the F1 case might have been infected by the F0 case to the time where the F1 case is placed under medical quarantine.
2. Definition of epidemic hotspots
2.1. Epidemic hotspot: an epidemic hotspot is the area (village, neighborhood, hamlet, etc.) where a confirmed case stays before symptom onset or before the sample testing positive for the virus is collected.
2.2. Inactive epidemic hotspot: an epidemic hotspot is considered inactive when there are no new confirmed cases for 28 days after the last case is isolated.
2.3. Sample collection, preservation and transport: see Appendix 1 enclosed therewith.
2.4. Reporting:
Daily reporting requirements:
- Provincial Centers for Disease Control and Prevention (CDCs), district-level medical centers and commune-level medical stations shall manage lists of F0 cases, suspected cases, F1 cases, F2 cases and quarantined persons in areas under their management (including all local treatment facilities) and monitor their health condition; provincial CDCs shall submit consolidated reports on data using Form 7 in Appendix 5 and lists of F0 cases and suspected cases using Form 4 in Appendix 5 to Institutes of Hygiene and Epidemiology and Pasteur Institutes before 14:00 on a daily basis.
- Institutes of Hygiene and Epidemiology and Pasteur Institutes shall manage lists of F0 cases, suspected cases, F1 cases and quarantined persons in areas under their management and update information on their health condition; submit consolidated reports on data using Form 7 in Appendix 5 and lists of F0 cases and suspected cases using Form 4 in Appendix 5 to General Department of Preventive Medicine before 15:00 on a daily basis.
- Testing facilities, including public and private healthcare establishments and hospitals, must report on testing data (including test results, number of daily collected samples, number of completed tests, etc.) to provincial CDCs before 13:00 on a daily basis. Provincial CDCs shall send these reports to Institutes of Hygiene and Epidemiology and Pasteur Institutes before 14:00 on a daily basis. Institutes of Hygiene and Epidemiology and Pasteur Institutes shall submit consolidated reports on test results, number of daily collected samples, number of completed tests, etc. using Form 8 in Appendix 5 to General Department of Preventive Medicine before 15:00 on a daily basis.
- Institutes of Hygiene and Epidemiology and Pasteur Institutes shall regularly assess and analyze the COVID-19 situation, make forecasts and propose COVID-19 prevention and control solutions for areas under their management, and submit reports to General Department of Preventive Medicine before 14:00 of every Monday. General Department of Preventive Medicine shall submit consolidated reports to leaders of the Ministry and National Steering Committee for COVID-19 Prevention and Control (hereinafter referred to as “National Steering Committee”) before 17:00 on a daily basis.
- Testing facilities permitted to do confirmatory tests by the Ministry of Health shall update information on confirmed cases and test results to the system for automatic patient number assignment of the Ministry of Health, notify test results to sample senders and submit reports to the Department of Health, General Department of Preventive Medicine and local Institute of Hygiene and Epidemiology and Pasteur Institute.
- Testing facilities shall notify samples testing negative for SARS-CoV-2 to sample senders.
- Comply with reporting requirements for group-A infectious diseases according to regulations in Circular No. 54/2015/TT-BYT dated 28/12/2015 by the Ministry of Health providing guidance on reporting and declaration of infectious diseases.
III. PREVENTION MEASURES
1. Non-specific prevention measures
Proactively adopt the following prevention measures:
- Do not visit infected areas. Comply with the 5K requirements (mask - health declaration - no gathering - distancing - disinfection); when visiting crowded places, take personal protective measures such as mask use, hand hygiene, distancing, etc.
- Avoid direct contact with people having acute respiratory diseases (fever, cough, breathing difficulty); where it is necessary to have contact, wear a medical mask properly and keep a distance of at least 2 meters.
- People with suspicious symptoms must immediately notify the nearest healthcare facility for timely advice, examination and treatment. Call the healthcare facility first before visiting to inform it of the symptom(s) and recent travel history to receive appropriate support; do not visit crowded places. Students and workers with suspicious symptoms must take leave from school/work and notify health authority immediately.
- Keep personal hygiene and wash hands regularly under running tape with soap for at least 40 seconds or with regular sanitizer (containing at least 60% alcohol) for at least 20 seconds; rinse the mouth and throat with mouthwash and avoid touching the eyes, nose or mouth to prevent infection.
- Cover the nose and mouth when coughing or sneezing, preferably with a towel, handkerchief, disposable tissue paper or shirt sleeve to prevent respiratory fluids from spreading; wash hands with soap and water or sanitizer immediately after coughing or sneezing. Do not spit outside of designated places in public.
- Ensure food safety and only consume properly cooked food.
- Keep warm and improve health via proper diet, resting and physical exercise.
- Enhance ventilation in housing and at workplaces by opening windows and doors; avoid using air-conditioners.
- Regularly clean living space, workplaces, schools, factories, etc. by wiping the floor, doorknobs and item surfaces using soap, regular cleaning agents or other disinfectants according to guidelines of the healthcare sector.
- Regularly clean and disinfect transport vehicles such as aircrafts, trains, watercrafts, automobiles, etc.
2. Specific prevention measures
Receive a COVID-19 vaccine according to indication and guidance for prevention as per regulations.
3. Border health quarantine
- Supervise persons entering Vietnam and implement regulations on health declaration according to the Government’s Decree No. 89/ND-CP dated June 25, 2018 providing guidelines for implementation of the Law on Prevention and Control of Infectious Disease regarding the border health quarantine, directions of National Steering Committee and guidelines of the Ministry of Health.
- Quarantine and medical handling at checkpoints shall be carried out according to regulations in the Government’s Decree No. 101/2010/ND-CP dated September 30, 2010 on guidelines for Law on Medical Examination and Treatment in terms of implementation of isolation measures, enforced isolation measures and specific anti-epidemic measures during epidemic period, directions of National Steering Committee and guidelines of the Ministry of Health.
4. Drugs, materials, chemicals and equipment for COVID-19 prevention and control
Provincial governments shall proactively prepare sufficient drugs, materials, chemicals and equipment for COVID-19 monitoring, testing, prevention and control in their provinces following the 4 on-sites motto.
IV. CONTROL MEASURES
A. General measures
1. F1 and F2 investigation and tracing
- This task must be done in a rapid and thorough manner. Do not miss any case. Provincial and district-level steering committees for COVID-19 prevention and control and relevant regulatory bodies and units shall mobilize all resources to ensure fast and efficient tracing.
- Start tracing as soon as possible after receiving information on the case.
Carry out tracing according to the rules and methods in the “Contact Tracing for COVID-19 Handbook” of the Ministry of Health.
2. Sample collection for COVID-19 control
- Collect samples from people in the epidemic hotspot for COVID-19 control and hotspot assessment and monitoring.
- Collect samples for SARS-CoV-2 testing from people and areas facing high risk outside of the hotspot to assess general community situation.
- According to the COVID-19 situation and risk assessment, National Steering Committee, Ministry of Health or provincial steering committees for COVID-19 prevention and control (hereinafter referred to as “provincial steering committees”) shall revise testing strategies, testing scale and sample collection procedures as appropriate.
3. Medical isolation/quarantine and handling
3.1. Confirmed cases
- Admit, isolate, manage and treat COVID-19 patients according to guidelines of the Ministry of Health.
- Avoid patient referral to prevent infection, unless the patient’s condition is beyond the treatment capacity of the facility.
3.2. F1 cases
- Quarantine all F1 cases in centralized quarantine facilities for at least 14 days after the last time they have contact with a confirmed case. It is recommended to establish separate centralized quarantine facilities for F1 cases as they face higher infection risk than other quarantined groups. If a separate centralized quarantine facility is not available, arrange for F1 cases to stay in a separate area. Persons of the same households, living in the same houses or working in same rooms as confirmed cases must be quarantined separately from other people as they face the highest infection risk. Assign persons having similar epidemiological characteristics and/or arriving at the facility at the same time to the same quarantine rooms/quarantine areas.
- Collect samples for immediate Real time RT-PCR testing for SARS-CoV-2 at least twice during the quarantine period.
- Collect the first sample immediately after the person is quarantined.
- Collect the second sample on the 14th day after the person is quarantined.
+ If the person tests positive for SARS-CoV-2, treat them as an F0 case.
+ If the person tests negative for SARS-CoV-2 at least twice, they may stop quarantining and shall start self-monitoring their health condition in their lodging for the next 14 days, not participate in mass gatherings, and follow the 5K requirements.
For persons entering Vietnam and required to undergo centralized quarantine for at least 14 days from the date on which quarantine starts, they shall be quarantined, have their samples collected and tested as F1 cases.
In case there are a large number of F1 cases beyond the centralized quarantine capacity or other special cases (the elderly, people with movement difficulties, children, persons requiring care, etc.), consider quarantining F1 cases in their lodgings for 14 days according to specific guidelines from the Ministry of Health and/or provincial steering committees. Collect samples on the first day and the last day of quarantine; if a person tests negative for SARS-CoV-2 at least twice, they may stop quarantining in their lodging and shall self-monitor their health condition in their lodging for the next 14 days, not leave their lodging without notifying local government, not join mass gatherings, and follow the 5K requirements strictly.
3.3. F2 cases
- Draw up F2 case lists and collect samples from F2 cases based on risk and epidemiological assessment.
- Quarantine F2 cases in their lodgings while waiting for Real time RT - PCR test results of the F1 case.
+ If the first sample of the F1 case tests positive for SARS-CoV-2, treat F2 cases as F1 cases.
+ If the first sample of the F1 case and first samples of F2 cases (if any) test negative for SARS-CoV-2, based on risk and epidemiological assessment, determine which F2 cases may stop quarantining in their lodgings and shall self-monitor their health conditions for 14 days, and notify health authority and community COVID team if developing any suspicious symptom.
3.4. Suspected cases
a. Suspected cases found in the community
- Request the suspected case to wear a mask and undergo temporary quarantine at home/in lodging immediately. The suspected case and their family must comply with the 5K requirements strictly. Do not have contact with family members and other people.
- Local health authority shall collect a sample for immediate Real time RT-PCR testing for SARS-CoV-2:
+ If the suspected case tests negative for SARS-CoV-2, they do not have COVID-19.
+ If the suspected case tests positive for SARS-CoV-2, treat them as an F0 case.
b. Suspected cases found in healthcare facilities
- Screen the suspected case according to regulations of the treatment system and place the suspected case under temporary quarantine in a separate room in an area away from the area for F0 cases and other treatment areas of the healthcare facility.
- Collect a sample for immediate Real time RT-PCR testing for SARS-CoV-2:
+ If the suspected case tests negative for SARS-CoV-2, they do not have COVID-19.
+ If the suspected case tests positive for SARS-CoV-2, treat them as an F0 case.
3.5. Persons having epidemiological connections with confirmed cases in other situations
For persons who have no close contact with a confirmed case but are present in a large mass event or on a transport vehicle with a confirmed case, health authority shall notify these persons by phone, message, social network or other mass media so that they can contact local health authority to receive instructions on health declaration, health monitoring, quarantine and testing as appropriate to epidemiological investigation results or at the request of the provincial steering committee.
4. Organization of community-based COVID-19 control
Launch a movement to encourage all people to join the fight against COVID-19 and establish community COVID teams in all residential areas that operate robustly and efficiently as follows:
- Every community COVID team shall consist of 2-3 people who are officials of the neighborhood/village, socio-political organizations and mass organizations and volunteers of the residential area. Depending on actual circumstances, each team shall take charge of 30-50 households with detailed household list.
- Duties: visit every street and house on a daily basis to:
+ Encourage and remind people to take measures against COVID-19 in their households. Request and instruct people to monitor health condition and check body temperature of all household members (if having a thermometer). Provide phone number and request people to proactively make health declaration when they or a family member develop/s fever, cough or other suspicious symptoms.
+ Ask, supervise, detect and immediately report suspected cases found in households with fever, cough, throat pain, influenza, fatigue, respiratory tract infection, etc. by phone to local government and commune-level health authority to have these cases quarantined and their samples collected promptly. Grasp the situation and risk factors in each household.
+ Detect and report persons refusing to make health declaration or comply with measures against COVID-19 according to regulations; persons entering Vietnam illegally, persons coming from infected areas, etc. to competent authorities.
+ Assist local government and health authority in F1 and F2 tracing when there is a case in an area under its management.
+ Perform other suitable tasks assigned by commune-level steering committees for COVID-19 prevention and control.
- COVID-19 prevention for community COVID teams: members of community COVID teams must always wear masks, use hand sanitizer and wear face shields (if any) while performing their duties. When performing their duties, community COVID teams may not enter people's houses; and shall knock on their doors, stand outside of the houses, request people to wear masks and keep a distance as far as possible to prevent infection (at least 2 meters apart).
Community COVID teams should create Zalo groups for households under their management for daily reporting and contact.
5. Environmental decontamination
- Carry out environmental decontamination where hotspots are located according to Appendix 4 enclosed therewith.
- Persons performing environmental decontamination must wear sufficient PPE while they work.
- After environmental decontamination, request the residents to refrain from moving for at least 30 minutes to ensure decontamination efficiency.
- All F0 transporting vehicles shall be disinfected with 0,1% active chlorine solution.
- Prioritize surface disinfection by wiping frequently touched areas such as table and chair surfaces, bed frames, wardrobes, refrigerators, doorknobs, handrails, sinks, restrooms, faucets, kitchen shelves, house walls, windows, doors, etc. of houses, workplaces, factories, schools, etc.. In case wiping is not possible, use shoulder-worn or hand-operated sprayers; do not spray on humans directly. Disinfection of other relevant areas by surface spraying shall be decided by epidemiological officials and health environment officials based on actual investigation, ensuring that all contaminated areas, areas suspected to be contaminated and areas posing risk of community transmission are decontaminated. Number of decontamination times shall be decided based on actual state of contamination in hotspots.
Based on the COVID-19 situation, epidemiological, virological and clinical research and investigation results, and recommendations of WHO, the Ministry of Health will revise these guidelines as appropriate to actual situation.
6. Communication about COVID-19 prevention and control
Properly follow the 5K requirements (mask - health declaration - distance - no gathering - disinfection) and get vaccinated against COVID-19. Request use of masks on public transit and in public places and crowded places. Limit mass events, practice safe distancing in public. Communicate measures against COVID-19 via different means in public and to each household. Encourage people to fulfill their responsibilities as citizens and responsibilities to the society for COVID-19 prevention. Launch a movement to motivate everyone to join the fight against the pandemic.
B. Specific measures for handling of epidemic hotspots upon detection of COVID-19 community transmission
1. Rules: regard COVID-19 control speed as the most important factor; ensure rapid tracing, rapid quarantine, rapid hotspot identification, and rapid sample collection and testing.
2. Specific handling measures are provided in Appendixes 1 to 5 enclosed therewith.
APPENDIX 1
HANDLING OF EPIDEMIC HOTSPOT UPON DETECTION OF COVID-19 COMMUNITY TRANSMISSION
(Enclosed with Decision No. … dated …/…/2021 by Ministry of Health)
1. Rules:
Regard COVID-19 control speed as the most important factor; ensure rapid tracing, rapid quarantine, rapid hotspot identification, and rapid sample collection and testing.
2. Specific activities:
- Immediately place F0 cases under isolation, and manage and provide them with treatment according to regulations.
- Carry out epidemiological investigation following normal rules.
- Find, quarantine and collect samples from F1 cases.
- Find, quarantine and collect samples from F2 cases (if necessary).
- Immediately impose temporary quarantine on the residential area with the COVID-19 case. Quarantine scope shall be decided based on initial assessment of the hotspot’s situation and risk. Such temporary quarantine may be imposed regardless of administrative boundaries and does not require an administrative decision.
- Depending on the hotspot’s size, make an epidemiological assessment to collect samples extensively from the temporarily quarantined area;
Collect samples extensively from the temporarily quarantined area as fast as possible. It is recommended to pool samples from the same household or households living close to each other. Test pooled samples immediately to assess the risk and situation of community transmission.
- Place the hotspot under quarantine as follows:
+ Based on testing results of F1 cases and community samples from the hotspot, decide the official quarantine scope. Rules for hotspot quarantine: quarantine neatly according to how far the risk runs (risk shall be assessed based on locations of F0 cases, F1 cases and exposure sites; and epidemiological connections in the community). Adopt the “Handbook for quarantine practices in areas with reported confirmed COVID-19” of the Ministry of Health in the quarantined area.
+ 2 objectives of hotspot quarantine: strictly control the hotspot to prevent the disease from spreading to other areas and thoroughly eliminate the virus inside the hotspot to prevent infection inside the hotspot. To achieve these 2 objectives, the quarantined area must cease all entry and exit and impose strict quarantine between houses inside the area according to the following rules: people should stay inside their houses; not meet anyone outside; not visit other houses; not let anyone visit their houses; and all stores should be closed. Set up entry and exit checkpoints for the quarantined area.
+ Collect samples every three days from all people in the quarantined area and pool samples from the same household or households living close to each other to promptly detect and isolate F0 cases from the community, eliminating the hotspot. Decide frequency of sample collection based on testing results. Hotspots with complicated community transmission require high sample collection frequency to eliminate the pathogens completely.
+ Publish a consolidated list of local quarantined areas and areas recording community transmission cases on the website of the province or city; regularly update the abovementioned list (contact [email protected] for guidance and edit access). To give recommendations on travel and quarantine of persons from quarantined areas and areas recording community transmission cases, the Ministry of Health has published lists of quarantined areas and areas recording community transmission cases on the https://moh. gov. vn/ website.
- Consider quarantine according to Directive No. 15/CT-TTg dated 27/3/2020 or Directive No. 16/CT-TTg dated 31/3/2020 by the Prime Minister or implement some basic content of these Directives in the commune recording the COVID-19 case, some nearby communes or communes with an important exposure site or epidemiological connection and adopt other directions of the Government, National Steering Committee and provincial steering committee.
- Organize comprehensive monitoring of and collect samples from all persons having fever, cough, throat pain, influenza and/or respiratory tract infection in the community and local treatment facilities and pharmacies. For the first days, check and collect samples from all persons having fever, cough, throat pain, fatigue, respiratory tract infection and/or loss of taste or smell in the area.
- Establish community COVID teams in all residential areas. Community COVID teams must operate vigorously and efficiently, visit every street and house to perform their duties, and request household representatives to make daily health declaration via their phones with community COVID teams or local health authority.
- Raise the awareness of every residential area and encourage all citizens to join the fight against COVID-19 via a movement, especially about complying with the 5K requirements, social distancing measures currently implemented and directions of the Government, National Steering Committee and provincial steering committee.
- Local government shall direct police and neighborhood watch forces to inspect and supervise compliance with COVID-19 control requirements of people in the infected area. Impose strict penalties upon those failing to abide by regulations to ensure that people properly follow COVID-19 control regulations.
- Carry out environmental decontamination where the F0 case lives and works according to Appendix 3 enclosed therewith.
Some notes:
- Temporary quarantine and collection of samples from the community should be done on a large scale but quarantine should be officially imposed according to how far the risk runs.
- It is advised that the first test for cases with suspicious symptoms and F1 cases is performed with individual samples;
- Samples from the community should be pooled to save biologicals (following Decision No. 1817/QD-BYT dated April 7, 2021 introducing interim guidelines for SAR-CoV-2 pool testing).
APPENDIX 2
SAMPLE COLLECTION, PRESERVATION AND TRANSPORT
(Enclosed with Decision No. 3638 dated 30/07/2021 by Ministry of Health)
1. Samples
Samples from suspected COVID-19 cases must be collected by healthcare workers or persons who have received training in sample collection. At least 01 respiratory tract sample must be collected for each case.
1.1. Genetic material detection testing
a. Upper respiratory tract samples:
+ Nasopharyngeal swab;
If nasopharyngeal swab cannot be collected, collect one of the following types of sample:
+ Throat swab;
+ Nasal swab (both nostrils);
b. Lower respiratory tract samples:
+ Sputum;
+ Tracheal aspirate, broncheoalveolar lavage (BAL) fluid, pleural fluid, etc.;
+ Lungs, bronchi, trachea.
1.2. Antigen detection testing
+ Nasopharyngeal sample;
+ Throat swab;
+ Nasal swab (both nostrils);
1.3. Antibody detection testing
+ Blood sample (not required, depending on serological testing, local governments and units shall formulate detailed plans)
+ Blood sample volume: 3ml - 5ml
2. Sampling methods
2.1. Tool preparation
- Tools for nasopharyngeal swab, throat swab and nasal swab collection must not have a calcium or wooden handle; synthetic-tipped sticks are preferred.
- Tongue depressors;
- 15ml conical centrifuge tube containing 2-3ml of viral transport medium;
- Plastic container (Falcon 50ml tube) or nylon bag for sample packaging;
- Gauzes soaked in antiseptic;
- Antiseptic alcohol, pens
- Waterproof PPE (separate-piece or one-piece) or long-sleeved medical waterproof gown covering from the neck to the knees and tied behind the neck and around the waist.
- Safety goggles;
- Clean powder-free gloves;
- Medical masks with high filtration rate (N95 or equivalent);
- Sterile 10 ml syringes;
- Sterile tubes without anticoagulant.
- Cold sample preserving containers.
2.2. Procedure
2.2.1. Wearing PPE
Step 1: Wash hands.
Step 2: Wear boots/shoe covers.
Step 3: Wear long gown or suit (separate-piece or one-piece).
Step 4: Wear mask.
Step 5: Wear safety goggles (goggles with temples).
Step 6: Wear cap and make sure that it completely covers hair, head, ears and mask strings.
Step 7: Wear face shield or safety goggles (goggles with strings to be worn outside of cap).
Step 8: Wear clean powder-free gloves.
Note: it is not necessary to wear both goggles and face shield.
2.2.2. Sample collection method for genetic material detection testing
2.2.2.1. Method for nasopharyngeal swab and throat swab collection
a) Method for nasopharyngeal swab collection
- Ask the patient to sit still, sneeze nasal discharge into tissue paper, and tilt their head slightly backwards; children must be held by adults.
- Tilt the patient’s head back 70° and support the back of the patient’s neck with one hand.
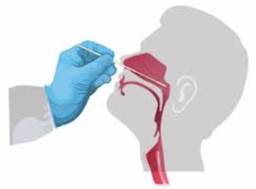
- Use the other hand to gently insert the swab into the patient’s nose and rotate the swab at the same time to ease the slide until reaching half the distance from the corner of the nose to the front of the ear.
Note: if resistance is met before reaching such depth, remove the swab and try the other nostril. When reaching the posterior nasopharynx, stop, rotate the swab and slowly remove it.
- Keep the swab in place for 5 seconds to ensure maximum absorbency.
- Slowly rotate and remove the swab.
- Put the tip of the swab in a vial containing transport medium and break the shaft of the swab at the score mark to fit it in the vial. Note: the nasopharyngeal swab and the throat swab shall be put together in one vial.
- Recap the vial tightly and wrap it in paraffin paper (if any).
Note: Children shall sit in their parents’ laps with their backs to their parents' chests. The parent shall hold the child’s body and hands tightly and tilt the child’s head back.
|
|
Insert the sterile swab in a straight line into one nostril (do not tilt upwards) along the nasal floor until reaching the pharynx. |
Picture 1: nasopharyngeal swab collection
b) Method for throat swab collection
- Ask the patient to open their mouth wide.
- Use a tongue depressor to hold the patient’s tongue down.
- Insert the swab into the throat, rub and rotate the swab 3-4 times against both tonsils and the posterior pharyngeal wall to obtain secretion and cells from the throat.
- Place the swab in a vial containing 3ml of transport medium (VTM or UTM) for preservation. Note that the tip of the swab must be completely submerged in the transport medium. If the swab is longer than the vial, cut or break its handle to fit it inside the vial.
- Recap the vial tightly and wrap it in paraffin paper (if any).
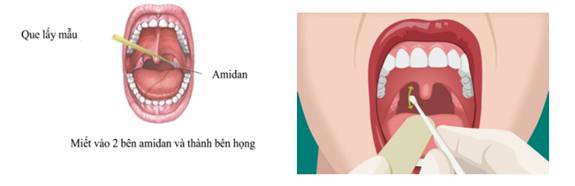
Picture 2: throat swab collection
2.2.2.2. Method for nasal swab collection
- Ask the patient to sit still and sneeze nasal discharge into tissue paper; children must be held by adults.
- Tilt the patient’s head slightly backwards and support the back of the patient’s neck with one hand.
- Use the other hand to gently insert the swab 2 cm into the patient’s nose and rotate the swab against the nasal wall for 3 seconds. After finishing with one nostril, use the same swab to collect sample from the other nostril.
- Put the tip of the swab in a vial containing transport medium and break the shaft of the swab at the score mark to fit it in the vial.
- Recap the vial tightly and wrap it in paraffin paper (if any).
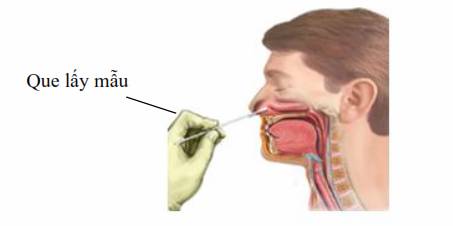
Picture 3: nasal swab collection
2.2.2.3. Broncheoalveolar lavage fluid
This type of sample is collected from patients mechanically ventilated via an endotracheal tube. Place a suction catheter along the endotracheal tube and use a pump to collect the broncheoalveolar lavage fluid into a vial containing transport medium.
2.2.3. Samples for antigen testing shall be collected and preserved according to manufacturer’s guidelines.
2.2.2.4. Blood samples for antibody detection testing
Use a sterile syringe to collect 3ml-5ml venous blood into a serum separator tube without anticoagulant and store it at 2°C - 8°C for up to 48 hours. If preserved longer, samples must be stored at -70°C.
Note:
- Include name, age and address of patient, type of sample and date of collection on sample vials.
- Lower respiratory samples (tracheal aspirate, broncheoalveolar lavage (BAL) fluid, pleural fluid) must be collected with the participation of the doctor(s) in charge of the patient.
2.2.3. Removing PPE
2.2.3.1. Removing long gown, cap and shoe covers (if they are separated from body suit)
Step 1: Remove gloves, turn them inside out and discard in waste container.
Step 2: Wash hands.
Step 3: Untie and remove gown, turn it inside out, roll into a bundle and discard in waste container.
Step 4: Wash hands.
Step 5: Remove shoe covers, turn them inside out and discard in waste container. Step 6: Wash hands.
Step 7: Remove face shield or safety goggles (for goggles with strings worn outside of cap).
Step 8: Wash hands.
Step 9: Remove hood by sliding hands inside it. Step 10: Remove safety goggles (goggles with temples and strings worn inside cap).
Step 11: Remove mask (using strings behind head or ears).
Step 12: Wash hands.
Note: PPE shall be worn and removed in two separate areas.
2.3.2.2. For separate pants, gown and cap
Step 1: Remove gloves, turn them inside out and discard in waste container.
Step 2: Wash hands.
Step 3: Untie and remove gown, turn it inside out, roll into a bundle and discard in waste container.
Step 4: Wash hands.
Step 5: Remove pants and shoe covers at the same time, turn pants inside out, roll into a bundle and discard in waste container.
Step 6: Wash hands.
Step 7: Remove face shield or safety goggles (for goggles with strings worn outside of cap).
Step 8: Wash hands.
Step 9: Remove hood by sliding hands inside it.
Step 10: Remove safety goggles (goggles with temples and strings worn inside cap).
Step 11: Remove mask (using strings behind head or ears).
Step 12: Wash hands.
Note: PPE shall be worn and removed in two separate areas.
2.2.3.2. Removing protective suit with attached hood
Step 1: Remove gloves, turn them inside out and discard in waste container.
Step 2: Wash hands.
Step 3: Remove face shield or safety goggles with strings worn outside of cap. If strings are worn inside of cap, remove cap before goggles.
Step 4: Wash hands.
Step 5: Remove hood and suit, turn them inside out, roll into bundle and discard in waste container.
Step 6: Wash hands.
Step 7: Remove shoe covers, turn them inside out and discard in waste container.
Step 8: Wash hands.
Step 9: Remove mask (using strings behind head or ears).
Step 10: Wash hands.
Note: PPE shall be worn and removed in two separate areas.
2.2.3.3. Disinfection of sampling equipment and collection sites
- Put all PPE and dirty tools in a heat-resistant biomedical waste nylon bag (use new gloves and mask).
- Tie and dry the bag at 120°C for 30 minutes before it may be collected and treated with other biomedical wastes as per regulations.
- Wash hands with soap; wipe and disinfect all tools, sample collection site and cold containers used to transport samples to laboratory with chloramine 0,1%.
3. Preservation, packing and transport of samples to laboratory
3.1. Preservation
Samples must be delivered to a laboratory as soon as possible and:
- Samples shall be stored at 2°C-8°C and delivered to the laboratory as soon as possible within 48 hours after collection. If samples cannot be delivered within 48 hours after collection, they must be stored at -70°C.
- Do not store samples in freezers or at -20°C.
3.2. Sample packing
- Samples shall be packed in three layers according to standards set by the Ministry of Health in Circular No. 40/2018/TT-BYT.
- Check to ensure that information on each sample vial and information in the investigation form are consistent with each other.
- Check to ensure that sample vials are properly capped and wrapped in paraffin (if any) or blotting paper.
- Place sample vials in a waterproof bag/nylon bag or container with lid and close it tightly.
- Place the bag/container containing sample vials in a cold container or hard shell box.
- Add ice-packs to the sample container to ensure that samples are stored at 2-8°C during transport.
- For frozen samples, add ice-packs taken from -70°C freezers to ensure that samples do not thaw during transport.
- Test requests shall be placed in waterproof bags/other nylon bags (do not place them together with samples) inside the sample container, which shall be labeled according to regulations in Circular No. 40/2018/TT-BYT.

3.3. Transport of samples to laboratory
- Samples must be stored at 2-8°C (or -70°C for frozen samples) during transport.
- Test requests and investigation forms must be sent together with samples.
- The sample sender shall immediately notify the laboratory of the estimated time of arrival of the samples so that the laboratory’s officials can prepare to receive the samples.
- Choose the fastest transport means and ensure that samples are properly preserved during transport.
APPENDIX 3
GUIDELINES ON USE OF CHLORINE-BASED DISINFECTANTS IN COVID-19 PREVENTION AND CONTROL
(Enclosed with Decision No. 3638 dated 30/07/2021 by Ministry of Health)
1. Introduction
Chlorine (Cl) is one of the halogens commonly used for disinfection due to their high biocide activity, which is facilitated by reduction-oxidation. When dissolved in water, these chemicals will release active chlorine to eliminate bacteria. Common chlorine chemicals include:
- Chloramine B containing 25% - 27% active chlorine.
- Chloramine T.
- Calcium hypochlorite.
- Sodium dichloroisocyanurate.
- Bleach (sodium hypochlorite or potassium hypochlorite).
2. Use of chemicals containing chlorine in epidemic prevention
- In infectious disease prevention, solutions produced from chemicals containing 0,05% and 0,1% active chlorine are usually used for different disinfection purposes and methods. A solution’s concentration must be calculated based on active chlorine.
- As different chemicals have different active chlorine contents, it is necessary to calculate amounts of chemicals needed to obtain a solution with desired active chlorine concentration.
- Amount of chlorine necessary to obtain a solution with desired active chlorine concentration is calculated using the following formula:
|
Amount of chlorine-releasing compound (gram) |
= |
Desired active chlorine concentration (%) x liter |
x |
1000 |
|
Active chlorine content of chlorine-releasing compound (%)* |
* Active chlorine content of the chlorine-releasing chemical is written on the product’s label, wrapping or instructions for use.
*Example:
- To obtain 10 liters of a solution containing 0,05% active chlorine from Chloramine B powder containing 25% active chlorine, the amount of Chloramine B powder needed is (0,05 x 10 / 25) x 1000 = 20 grams.
- To obtain 10 liters of a solution containing 0,05% active chlorine from calcium hypochlorite powder containing 70% active chlorine, the amount of calcium hypochlorite powder needed is (0,05 x 10 / 70 ) x 1000 = 7,2 grams.
- To obtain 10 liters of a solution containing 0,05% active chlorine from sodium dichloroisocyanurate powder containing 60% active chlorine, the amount of sodium dichloroisocyanurate powder needed is: (0,05 x 10 / 60) x 1000 = 8,4 grams.
Table 1. Amount of chlorine-releasing chemical necessary to obtain 10 liters of solution containing active chlorine commonly used in infectious disease prevention
|
Chemical name (active chlorine content) |
Amount of chemical necessary to obtain 10 liters of solution containing active chlorine |
|
|
0,05% |
0,1% |
|
|
Chloramine B 25% |
20g |
40g |
|
Calcium hypochlorite (70%) |
7,2g |
14,4g |
|
Sodium dichloroisocyanurate (60%) |
8,4g |
16,8g |
3. Method of mixing
- Fully dissolve necessary amount of chemical in 10 liters of clean water.
- As chlorine solutions lose strength over time, these solutions should be prepared in suitable quantity and used as soon as possible. It is best to prepare and use chlorine solutions within the day instead of preparing in advance. Prepared solutions shall be stored in closed containers in a dry, cool place out of direct sunlight.
Note:
- Chlorine-releasing compounds are effective against bacteria only when they are dissolved in water and become a solution (only then can they release biocidal active chlorine), thus, do not use chlorine-releasing compounds in powder form for disinfection.
- As chlorine solutions lose strength over time, these solutions should be prepared in suitable quantity and used as soon as possible. It is best to prepare and use chlorine solutions within the day instead of preparing in advance. Leftover solutions shall be stored in closed containers out of direct sunlight with a plan to use them as soon as possible.
APPENDIX 4
ENVIRONMENTAL DECONTAMINATION IN AREAS WITH COVID-19 COMMUNITY TRANSMISSION
(Enclosed with Official Dispatch No. 1560/BYT-MT dated 25/03/2020 by Ministry of Health)
1. Objectives
Prevent community transmission of COVID-19.
2. Rules
- Environmental decontamination must be promptly performed upon detection of the first COVID-19 case (F0 case).
- Prioritize surface disinfection by wiping frequently touched areas such as table and chair surfaces, bed frames, wardrobes, refrigerators, doorknobs, handrails, sinks, restrooms, faucets, kitchen shelves, house walls, windows, doors, etc. of houses, workplaces, factories, schools, etc.. In case wiping is not possible, use shoulder-worn or hand-operated sprayers.
- Surfaces and objects must be cleaned prior to disinfection.
- Ensure complete safety and prevent cross-infection to persons performing environmental decontamination.
3. Areas requiring environmental decontamination
- Next-door houses, rooms, apartments, apartment buildings, dorms, hotels, lodgings, centralized quarantine facilities, etc. (hereinafter referred to as “lodgings”) and workplaces, business establishments, industrial parks, etc. (hereinafter referred to as “workplaces”) of F0 cases;
- Areas surrounding lodgings and workplaces of F0 cases, including:
+ External walls of lodgings and workplaces of F0 cases;
+ Corridors, common corridors, stairs, elevators, lobbies, etc. of apartment buildings, dormitories, hotels, centralized quarantine facilities, workplaces, business establishments and industrial parks of F0 cases.
+ Walls, gardens, yards, sidewalks, shared walkways, etc. of adjacent houses.
4. Equipment and chemical preparation
- Cleaning and disinfection equipment; 2 buckets, 2 cleaning towels, mops, disinfectant spray bottles/machines, etc.
- Yellow biomedical waste bags/containers.
- PPE, including:
+ Medical masks for nose and mouth protection.
+ Goggles to protect ocular membranes from splash.
+ Protective clothing for protection against water and fluids.
+ Thick rubber gloves.
+ Waterproof boots or shoe covers.
- Disinfectants, including chlorine-based disinfectants; 70-degree alcohol; hand soap or hand sanitizers containing at least 60% alcohol.
5. Procedures
5.1. Inside lodgings and workplaces of F0 cases and inside next-door rooms/apartments
a) Cleaning and disinfection
Apply the 2-bucket wash method with one bucket filled with clean water and the other filled with a disinfectant solution containing 0.1% active chlorine. Use a clean towel for each wiping turn, do not rinse towels in the buckets and use each towel for an area no larger than 20 m2.
- Wipe frequently touched areas (table and chair surfaces, bed frames, wardrobes, refrigerators, doorknobs, handrails, sinks, restrooms, faucets, kitchen shelves, house walls, windows, doors, etc. of houses, workplaces, factories, schools, etc.) as follows:
+ Wipe the surfaces using a towel soaked with clean water.
+ Wipe the surfaces using towels soaked with disinfectant solutions from clean areas to dirty areas, from inside out and from the top to the bottom.
- Wipe the floors of the bedroom, living room, bathroom, kitchen, stairs, balcony, etc. as follows:
+ Wipe the floors with clean water first. If there is garbage, gather the garbage while wiping.
+ Mop the floors with disinfectant solutions from clean areas to dirty areas, from inside out and from the top to the bottom.
Some notes:
- Use 70-degree alcohol to wipe surfaces of electrical/electronic devices such as light switches, remotes, televisions, phones, etc. after turning off power supply.
- Replace the water or disinfectant solution when it is dirty.
b) Waste collection
All waste from houses of F0 cases must be treated as biomedical waste. Put waste in yellow bags, tie each bag tightly, put it inside another yellow bag and tie this bag tightly as well. All yellow bags must be labeled “CHẤT THẢI CÓ NGUY CƠ CHỨA SARS-CoV-2” (“COVID-19 DANGERS”).
c) Handling of reusable personal items (clothing, blankets, mosquito nets, tableware, etc.) of F0 cases
- Used textile items such as clothing, blankets, mosquito nets, bed linen and pillowcases of F0 cases must be fully soaked in a disinfectant solution containing 0.05% active chlorine for at least 20 minutes before washing.
- Tableware of F0 cases must be fully soaked in a disinfectant solution containing 0.05% active chlorine for 10 - 20 minutes before rinsing with clean water (if disinfectants are not available, tableware may be boiled for 10 - 15 minutes).
5.2. Outside lodgings and workplaces of F0 cases and inside next-door rooms/apartments
a) Chemicals: disinfectant solutions containing 0.1% active chlorine.
b) Wipe or spray chemical evenly at a rate of 0,3-0,5 liter/m2 in the following places:
- If the F0 case stays in an apartment building, dorm, hotel, lodging, centralized quarantine facility, etc.; and for the workplace of the F0 case:
+ Wipe or spray external walls of: i) room/apartment of the F0 case; ii) rooms/apartments next to the room/apartment of the F0 case.
+ Wipe shared areas, including i) corridors and walkways shared by the whole floor, row of rooms/apartments, rented room, factory, production team and/or work station; ii) staircases, elevators, lobby of the building/workplace; iii) other shared areas of the building/factory.
- If the F0 case lives in a private house:
+ For the house of the F0 case: wipe or spray chemical over the external walls, outside of windows and doors of the house; the entire yard, garden, kitchen and shared areas (if any) of the house;
+ For houses next to the F0 case’s house: wipe or spray chemical over the external walls, outside of windows and doors; yards, gardens, kitchens and shared areas (if any); spray chemical over walkways shared between the F0 and adjacent houses.
+ Spray chemical over public areas surrounding the F0 case’s house (if any) such as playgrounds, outdoor sports facilities, etc.
Some notes:
- Ask people to leave areas requiring disinfection before spraying chemical. After spraying chemical over shared areas, request people to refrain from moving for 30 minutes to ensure disinfection effectiveness.
- Change spraying locations based on how frequently the patient makes contact with their lodging and/or workplace.
6. Completing environmental decontamination
- Collect and put biomedical waste bags and used PPE into biomedical containers and handle them as regulated.
- Perform hand hygiene with soap and water or hand sanitizer containing at least 60% alcohol immediately after finishing environmental decontamination.
7. Carrying out environmental decontamination
- Departments of Health shall direct local health units to perform environmental decontamination.
- Local health units shall:
+ Prepare equipment and chemicals for environmental decontamination according to Section 4 of this Appendix;
+ Carry out environmental decontamination in areas with F0 cases under their management.
+ Collect and put biomedical waste bags and used PPE into biomedical containers.
- Managers of centralized quarantine facilities, lodgings, workplaces, etc. with F0 cases shall:
+ Inform quarantined persons about environmental decontamination for their cooperation.
+ Prepare the sites requiring environmental decontamination.
+ Cooperate with health units in environmental decontamination.
+ Contact environment units to have biomedical waste transported and handled with the day.